Assigning oxidation numbers9/7/2023 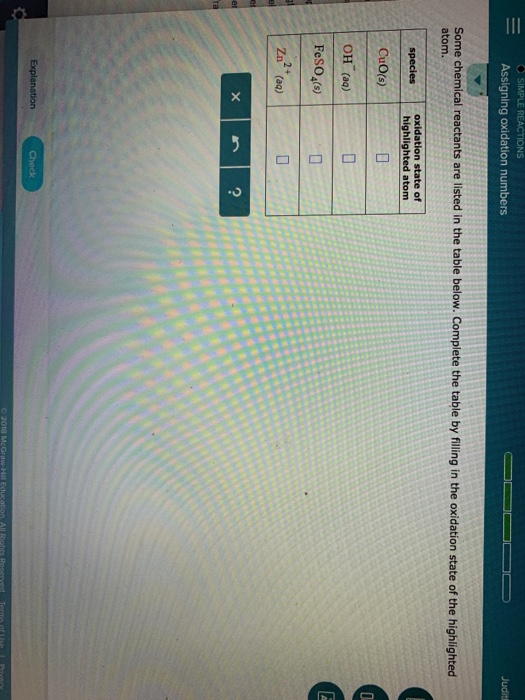
And what we wanna do in this video is think about which of theĮlements are being oxidized in this reaction and which of the elements are being reduced in this reaction. \(\).Have here is a reaction that involves iodine, However, this description in terms of electron loss and gain applies only to ionic compounds with a complete electron transfer and does not apply to some reactions, such as: Such identification may be made by knowing which substance is transferring electrons to other substances, according to the concept of redox reaction. 
In a chemical process, how do you identify the oxidised substance, the reduced substance, and the reducing and oxidising agents? Learn Oxidation Number & Oxidation State Here What are Oxidation Numbers? In this article, we will focus on the rules and calculation of oxidation numbers. An atom with higher electronegativity is given a negative oxidation state. It is defined as the charge that appears to form an ionic bond with other heteroatoms. The oxidation state helps us to ascertain the transfer of electrons. The oxidation number is the total number of electrons that atoms in a molecule can share, lose, or acquire while establishing chemical interactions with atoms of another element. In simple words, the oxidation number is the number assigned to the components in a chemical combination. When it comes to redox reactions, there are a few reasonably effective numbers, such as oxidation numbers. Oxidation Numbers:Numbers are very useful.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
